Key Takeaway
Blueroot Health Recalls Bottles of Bariatric Fusion Iron Multivitamins Due to Risk of Serious Injury or Death from Child Poisoning; Violates Mandatory Standard for Child-Resistant Packaging; Manufactured by VitaQuest International by VitaQuest International, LLC of Caldwell, New Jersey was recalled on September 11, 2025. What to do: Consumers should immediately secure the recalled bottles out of sight and reach of children, and contact Blueroot Health for information on how to obt...
Blueroot Health Recalls Bottles of Bariatric Fusion Iron Multivitamins Due to Risk of Serious Injury or Death from Child Poisoning; Violates Mandatory Standard for Child-Resistant Packaging; Manufactured by VitaQuest International
Description
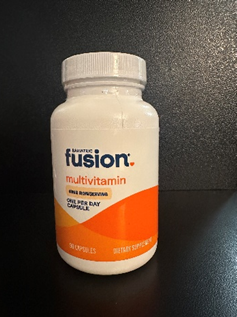
This recall involves two types of Bariatric Fusion dietary supplement bottles: high ADEK multivitamin capsules (90 and 270-count bottles) and One Per Day bariatric multivitamin capsules (90-count bottle), both with 45mg of iron. The recalled bottles are white and orange. Only bottles with smooth cap tops that lack the "push down & turn" embossed lettering are included in this recall. The Bariatric Fusion logo is printed on the front of the bottles. Lot number 0066J4, 0065J4, 0453B5 or 0370B5 is printed on the bottom of the bottles.
Injuries / Consequence
None reported
Remedy
Consumers should immediately secure the recalled bottles out of sight and reach of children, and contact Blueroot Health for information on how to obtain a free child-resistant replacement cap.
Products Affected
Bariatric Fusion Iron Multivitamin Bottles
Images


Company Information
VitaQuest International, LLC of Caldwell, New Jersey
Unknown location
View all 1 recalls by VitaQuest International, LLC of Caldwell, New Jersey →Frequently Asked Questions
Is this recall still active?
Check the status badge at the top of this page. "Ongoing" means the recall is still active and you should follow the remedy instructions. "Completed" or "Terminated" means the recall process has concluded, though the safety issue remains relevant for any unrepaired products.
How do I check if my product is affected?
Compare the model number, lot code, serial number, or date range listed in the recall notice with the information on your product's label or packaging. For vehicles, check your VIN at NHTSA.gov/Recalls.
What remedy is available for this recall?
The listed remedy for this recall is: Consumers should immediately secure the recalled bottles out of sight and reach of children, and contact Blueroot Health for information on how to obtain a free child-resistant replacement cap.
Where can I find the original recall notice?
This recall was issued by CPSC. You can view the original notice using the link provided above.