Key Takeaway
Dr. Reddy's Recalls Prescription Drug Blister Packages Due to Risk of Poisoning by Dr. Reddy's Laboratories, Ltd., of India was recalled on February 25, 2021. What to do: Consumers should immediately store the recalled medications in a safe location out of reach of children and contact Dr. Reddy's for a full refund.
Dr. Reddy's Recalls Prescription Drug Blister Packages Due to Risk of Poisoning
Description
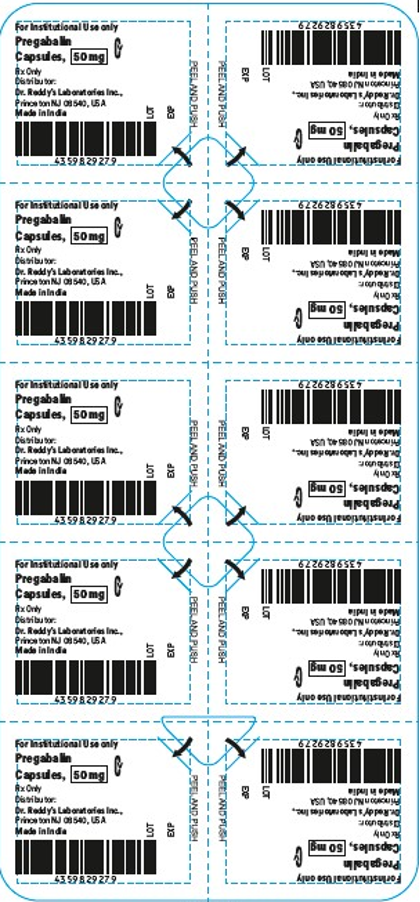
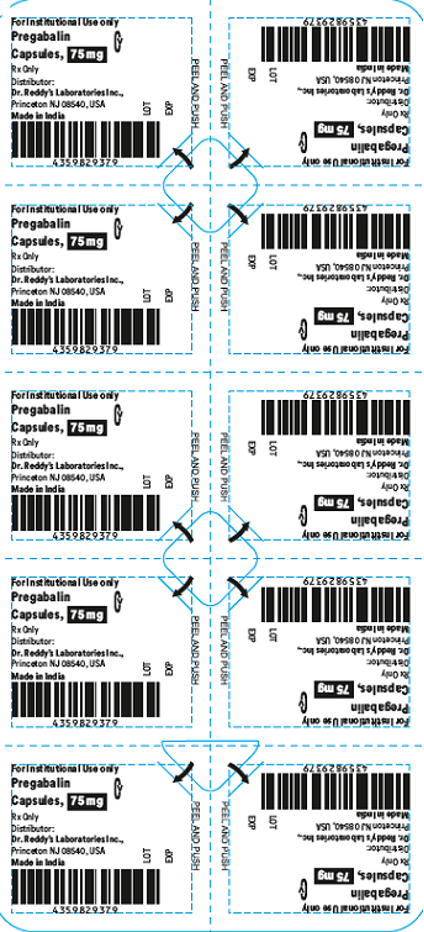
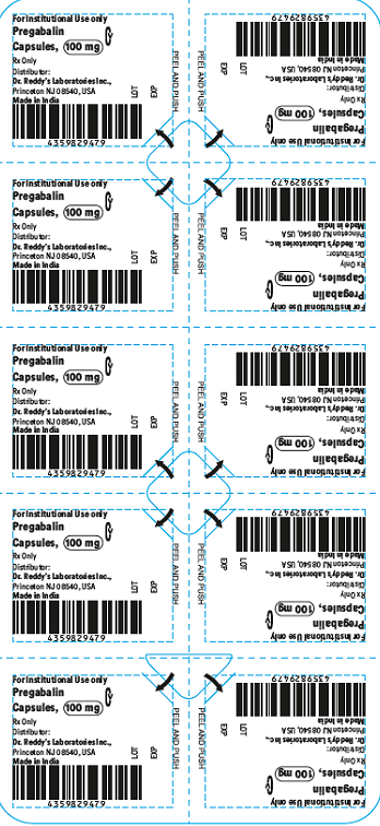
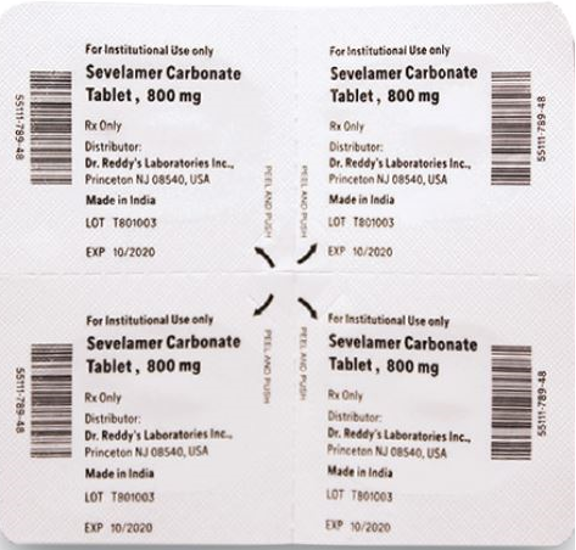
This recall involves blister packages of prescription medications. The name and strength of the medication, "For Institutional Use only," "Rx Only," lot number and expiration date are printed on the outside of the package as well as on the individual blister units. The Dr. Reddy's logo and NDC number are printed on the outside of the package. The recalled medications include the following: Recalled Prescription Drugs NDC Numbers Carton Configurations Lot Numbers Expiration Dates Imatinib Mesylate Tablets 100 mg 43598-344-31 3 blister cards of 10 tablets H2000138 2022-0630 Imatinib Mesylate Tablets 400 mg 43598-345-31 3 blister cards of 10 tablets H2000127 2022-0630 Pregabalin Capsules 50 mg 43598-292-66 5 blister cards of 10 capsules T900876 2021-0630 Pregabalin Capsules 75 mg 43598-293-66 5 blister cards of 10 capsules T901021 2021-0731 Pregabalin Capsules 100 mg 43598-294-66 5 blister cards of 10 capsules T901022 2021-0731 Pregabalin Capsules 150 mg 43598-295-66 5 blister cards of 10 capsules T901023 2021-0731 Sevelamer Carbonate Tablets 800 mg 55111-789-11 4 blister cards of 25 tablets T801003, T000009, T900221 2020-1031, 2021-1231, 2021-0228 Tadalafil Tablets 5 mg 43598-575-31 3 blister cards of 10 tablets T000376 2022-0131 Tadalafil Tablets 20 mg 43598-573-31 3 blister cards of 10 tablets T000425 2022-0228
Injuries / Consequence
No incidents or injuries have been reported.
Remedy
Consumers should immediately store the recalled medications in a safe location out of reach of children and contact Dr. Reddy's for a full refund.
Products Affected
Imatinib Mesylate Tablets 100 mg, Imatinib Mesylate Tablets 400 mg, Pregabalin Capsules 50 mg, Pregabalin Capsules 75 mg, Pregabalin Capsules 100 mg, Pregabalin Capsules 150 mg, Sevelamer Carbonate Tablets 800 mg, Tadalafil Tablets 5 mg and Tadalafil Tablets 20 mg
Images













Company Information
Dr. Reddy's Laboratories, Ltd., of India
Unknown location
View all 1 recalls by Dr. Reddy's Laboratories, Ltd., of India →Frequently Asked Questions
Is this recall still active?
Check the status badge at the top of this page. "Ongoing" means the recall is still active and you should follow the remedy instructions. "Completed" or "Terminated" means the recall process has concluded, though the safety issue remains relevant for any unrepaired products.
How do I check if my product is affected?
Compare the model number, lot code, serial number, or date range listed in the recall notice with the information on your product's label or packaging. For vehicles, check your VIN at NHTSA.gov/Recalls.
What remedy is available for this recall?
The listed remedy for this recall is: Consumers should immediately store the recalled medications in a safe location out of reach of children and contact Dr. Reddy's for a full refund.
Where can I find the original recall notice?
This recall was issued by CPSC. You can view the original notice using the link provided above.