Key Takeaway
First Aid Research Recalls Maximum Strength Bacitraycin Plus Ointment With Lidocaine Due to Failure to Meet Child Resistant Closure Requirement was recalled on March 7, 2018. What to do: Consumers should immediately place the recalled ointment out of the reach of children and contact United Exchange, the product's importer, for instruc...
First Aid Research Recalls Maximum Strength Bacitraycin Plus Ointment With Lidocaine Due to Failure to Meet Child Resistant Closure Requirement
Description
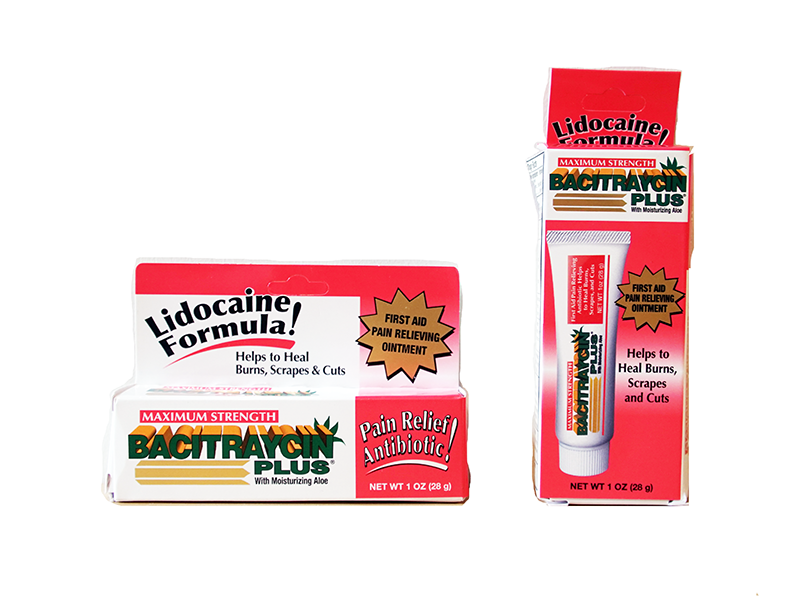
This recall involves Maximum Strength Bacitraycin Plus Ointment with Lidocaine. The recalled ointment is in a white, one ounce tube with "Bacitraycin Plus" printed on the front in green. Lidocaine is one of the two active ingredients listed on the back of the tube. The tube measures approximately 5 inches long by 1 inch wide. The lot number is printed on the end of the tube. The following lot numbers are included in the recall: Lot Numbers 16001 through 16002 404001 through 404002 405001 through 405003 406001 through 406004 407001 through 407002 415001 through 415010 416001 through 416003 417001 through 417004
Injuries / Consequence
None reported
Remedy
Consumers should immediately place the recalled ointment out of the reach of children and contact United Exchange, the product's importer, for instructions on how to receive a full refund from the place of purchase.
Products Affected
Maximum Strength Bacitraycin Plus Ointment with Lidocaine
Images

Frequently Asked Questions
Is this recall still active?
Check the status badge at the top of this page. "Ongoing" means the recall is still active and you should follow the remedy instructions. "Completed" or "Terminated" means the recall process has concluded, though the safety issue remains relevant for any unrepaired products.
How do I check if my product is affected?
Compare the model number, lot code, serial number, or date range listed in the recall notice with the information on your product's label or packaging. For vehicles, check your VIN at NHTSA.gov/Recalls.
What remedy is available for this recall?
The listed remedy for this recall is: Consumers should immediately place the recalled ointment out of the reach of children and contact United Exchange, the product's importer, for instructions on how to receive a full refund from the pla...
Where can I find the original recall notice?
This recall was issued by CPSC. You can view the original notice using the link provided above.