Key Takeaway
Sandoz Recalls Aprepitant Capsules and Lidocaine and Prilocaine Cream Prescription Drugs Due to Failure to Meet Child Resistant Packaging Requirement; Risk of Poisoning by Sandoz Inc., of Princeton, New Jersey (Lidocaine and Prilocaine cream) was recalled on March 9, 2023. What to do: Consumers should immediately secure the medications out of the sight and reach of children and contact Sandoz for a free child resistant pouch to stor...
Sandoz Recalls Aprepitant Capsules and Lidocaine and Prilocaine Cream Prescription Drugs Due to Failure to Meet Child Resistant Packaging Requirement; Risk of Poisoning
Description
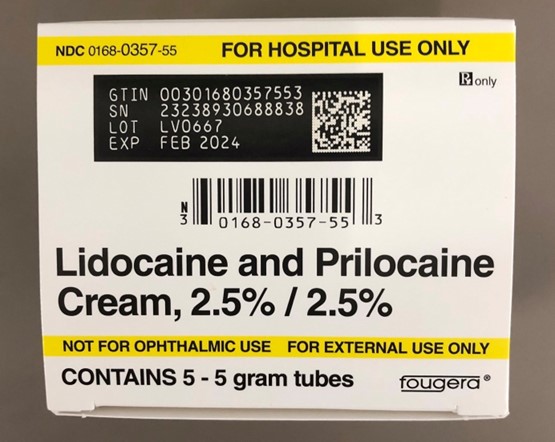
This recall involves prescription drugs Aprepitant 125 mg capsules sold in cartons containing one blister card of 6 capsules and 5 gram tubes of Lidocaine and Prilocaine cream sold in cartons containing 5 tubes and packed with or without 12 dressings. The Aprepitant capsules are in a non-child resistant blister card packaged in a carton that has the name "Sandoz," the name of the medication, dosage, NDC number, lot number, and expiration date on the carton and on the blister cards. The warnings "This unit-dose packaging is not child-resistant" and "For institutional use only" are listed on the carton. The Lidocaine and Prilocaine cream is packaged in a 5 gram tube with a continuous thread white closure. The name "fougera®," the name of the medication, dosage and NDC number are printed on the carton and tube and the expiration date and lot number are printed on the carton and stamped on the crimp of the tube. The warning "FOR HOSPITAL USE ONLY" is printed on the carton and the tube. Product Description NDC Number Lot Number Expiration Date Aprepitant Capsules 125 mg 0781-2323-68 Carton of 1 Blister Pack of 6 capsules 0781-2323-06 Blister Pack LK3209 LC6454 04/2024 12/2023 Lidocaine and Prilocaine 2.5%/2.5% Cream 5 gram Tubes 0168-0357-56 Carton of 5 tubes and 12 dressings 0168-0357-55 Carton of 5 tubes 0168-0357-05 Tube LA2782 LA2784 LV0667 LX5350 MA1640 MB3205 LA2785 LR9041 MB3209 03/2023 03/2023 02/2024 03/2024 03/2024 04/2024 03/2023 11/2023 04/2024
Injuries / Consequence
None reported
Remedy
Consumers should immediately secure the medications out of the sight and reach of children and contact Sandoz for a free child resistant pouch to store the products. Once the medication is secured, consumers can continue to use the medication as directed.
Products Affected
Aprepitant capsules and Lidocaine and Prilocaine cream
Images




Company Information
Sandoz Inc., of Princeton, New Jersey (Lidocaine and Prilocaine cream)
Unknown location
View all 1 recalls by Sandoz Inc., of Princeton, New Jersey (Lidocaine and Prilocaine cream) →Frequently Asked Questions
Is this recall still active?
Check the status badge at the top of this page. "Ongoing" means the recall is still active and you should follow the remedy instructions. "Completed" or "Terminated" means the recall process has concluded, though the safety issue remains relevant for any unrepaired products.
How do I check if my product is affected?
Compare the model number, lot code, serial number, or date range listed in the recall notice with the information on your product's label or packaging. For vehicles, check your VIN at NHTSA.gov/Recalls.
What remedy is available for this recall?
The listed remedy for this recall is: Consumers should immediately secure the medications out of the sight and reach of children and contact Sandoz for a free child resistant pouch to store the products. Once the medication is secured, co...
Where can I find the original recall notice?
This recall was issued by CPSC. You can view the original notice using the link provided above.