Key Takeaway
Sandoz Recalls Losartan Potassium and Ezetimibe Prescription Drug Bottles Due to Failure to Meet Child-Resistant Closure Requirements was recalled on August 29, 2019. What to do: Consumers should immediately secure the medications to keep them out of the sight and reach of children and contact Sandoz for a free replacement chil...
Sandoz Recalls Losartan Potassium and Ezetimibe Prescription Drug Bottles Due to Failure to Meet Child-Resistant Closure Requirements
Description
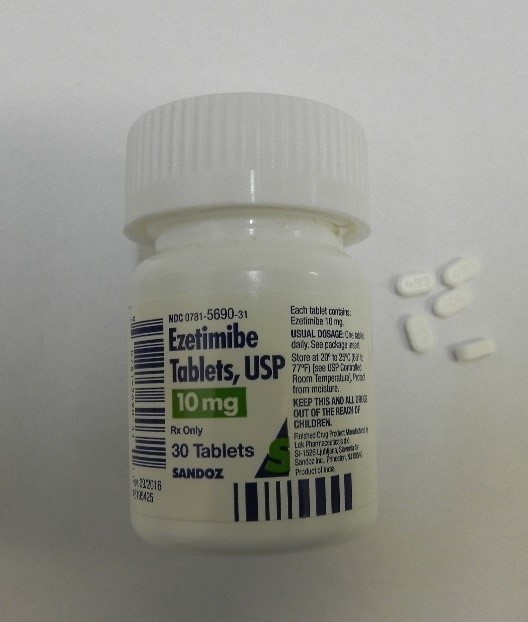
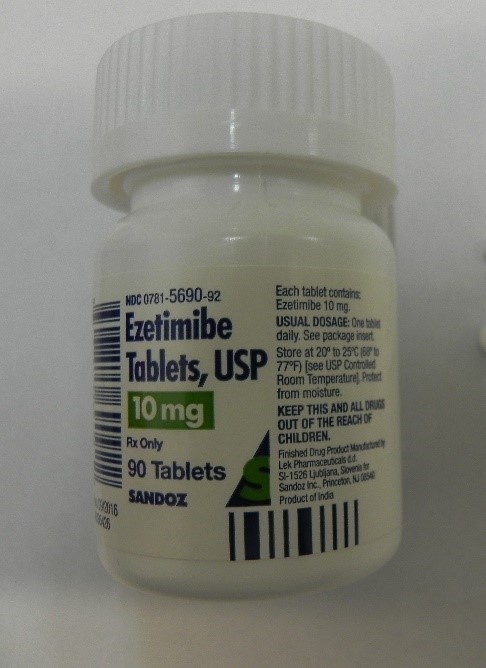
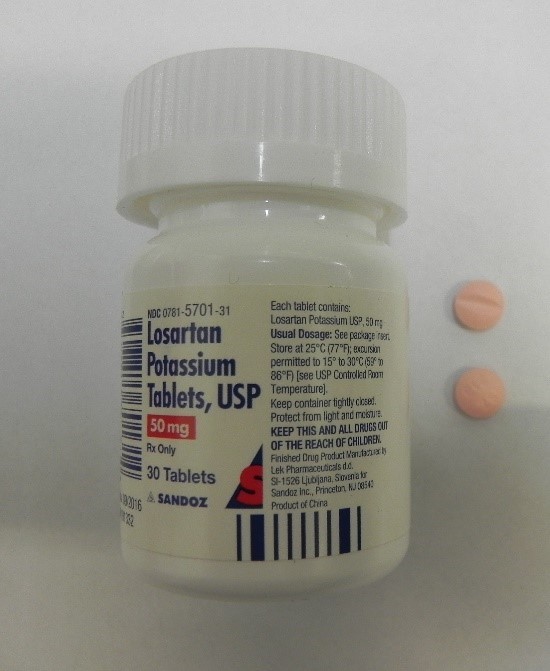
This recall involves bottles of prescription medications Losartan Potassium and Ezetimibe from Sandoz with certain lot numbers. The recalled bottles have "Sandoz," the name of the medication, dosage, and NDC on the front of the bottle labels and the lot number and expiration date on the side of the bottle labels. The recall includes the following: Recalled Prescription Drugs NDC Numbers Lot Numbers Expiration Date Ezetimibe 10mg Tablets 0781-5690-31 JE4491 Aug-2020 30 count bottle JE4492 Aug-2020 JE4493 Aug-2020 JE4495 Aug-2020 JG0308 Sep-2020 JG0310 Sep-2020 JG0311 Sep-2020 JG0312 Sep-2020 JG5061 Sep-2020 JG5063 Sep-2020 JK8921 Oct-2020 JK8922 Oct-2020 JK8923 Oct-2020 JK8924 Oct-2020 JL5535 Oct-2020 JM2253 Oct-2020 JM2254 Oct-2020 JM2255 Oct-2020 JM2257 Oct-2020 JM2258 Oct-2020 JM2259 Oct-2020 JM5986 Oct-2020 JM5987 Oct-2020 Ezetimibe 10mg Tablets 0781-5690-92 JE4481 Aug-2020 90 count bottle JG0249 Sep-2020 JK8989 Oct-2020 JN0764 Jan-2021 Losartan Potassium 50mg Tablets 0781-5701-31 HV9471 Feb-2020 30 count bottle
Injuries / Consequence
None Reported
Remedy
Consumers should immediately secure the medications to keep them out of the sight and reach of children and contact Sandoz for a free replacement child resistant bottle cap. Once the medication is secured, consumers can continue to use the medication as directed.
Products Affected
Losartan Potassium and Ezetimibe prescription drug bottles
Images

Frequently Asked Questions
Is this recall still active?
Check the status badge at the top of this page. "Ongoing" means the recall is still active and you should follow the remedy instructions. "Completed" or "Terminated" means the recall process has concluded, though the safety issue remains relevant for any unrepaired products.
How do I check if my product is affected?
Compare the model number, lot code, serial number, or date range listed in the recall notice with the information on your product's label or packaging. For vehicles, check your VIN at NHTSA.gov/Recalls.
What remedy is available for this recall?
The listed remedy for this recall is: Consumers should immediately secure the medications to keep them out of the sight and reach of children and contact Sandoz for a free replacement child resistant bottle cap. Once the medication is sec...
Where can I find the original recall notice?
This recall was issued by CPSC. You can view the original notice using the link provided above.